InterRett is the first-ever project collecting data on a global basis about Rett syndrome.
Funded by the International Rett Syndrome Foundation, this international online database examines the clinical features and genetic characteristics of Rett syndrome. InterRett is playing an invaluable role in:
- development of partnerships between families and clinicians and in advancing knowledge about Rett syndrome;
- increasing the clinical understanding of Rett syndrome;
- providing a new way to help families affected by the disorder, health professionals and the general public learn about Rett syndrome;
- encouraging collaboration with researchers from around the world.
Why InterRett is so special
For rare disorders such as Rett syndrome, the Internet provides access to a worldwide population, providing higher statistical power than individual centers or even country based research studies. The Internet also provides an ideal medium to disseminate high quality information about a specific disorder to the medical and general community.
Our mission
To collect and bring together data from many different and varied information sources to enhance our knowledge and build a clearer picture of Rett syndrome.
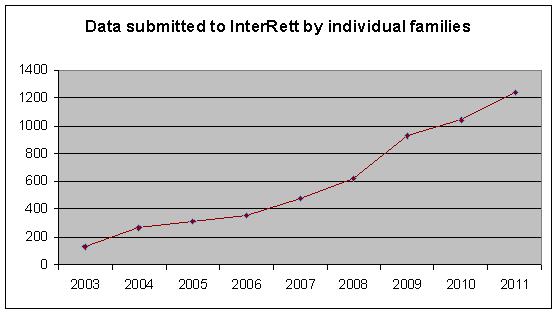
InterRett Data
Privacy
All data are de-identified (are assigned unique codes) to protect the privacy of participating families. To maintain the validity of the data all families and clinicians must register and receive login details before submitting data.
InterRett is managed and coordinated from the Australian Rett Syndrome Study at the Telethon Kids Institute (Perth, Western Australia) by Dr Helen Leonard, Professor Sue Fyfe (Curtin University) and Professor Nick de Klerk.
Where do we find information on individuals with Rett syndrome?
Information is sourced globally from:
- Rett syndrome clinical centres;
- clinician files;
- living experience of families and carers.
How do we bring it together?
- By providing easy access to standardised questionnaires to both professionals and families.
- Translating questionnaires into different languages.
- By providing custom databases and support to clinicians with multiple cases of Rett syndrome.
- Assisting with the conversion of hand-written notes into digital records.
- Contributing to the development of standardised protocols.
How are these data used?
- Searchable online database.
- Collaborative research with clinicians and researchers in Australia, China, France, Israel, Japan, Spain, UK and the USA.
Accessing InterRett data
Who has access to these data?
De-identified data is available to any researchers who meet the requirements of the Data Access Guidelines
Access to de-identified InterRett data is granted to bona fide researchers with the appropriate ethical approvals. It should be noted that researchers are responsible for their study design, funding and ethics approval.
Steps involved
- Discuss requirements with a member of the Data Management Committee.
- Complete an Application form
- Wait for approval
- Complete a Data Request Form
- Receive data
Also in this section...
InterRett Collaborating Clinicians
To see the full list of clinicians from around the world who have kindly contributed data to InterRett click here.